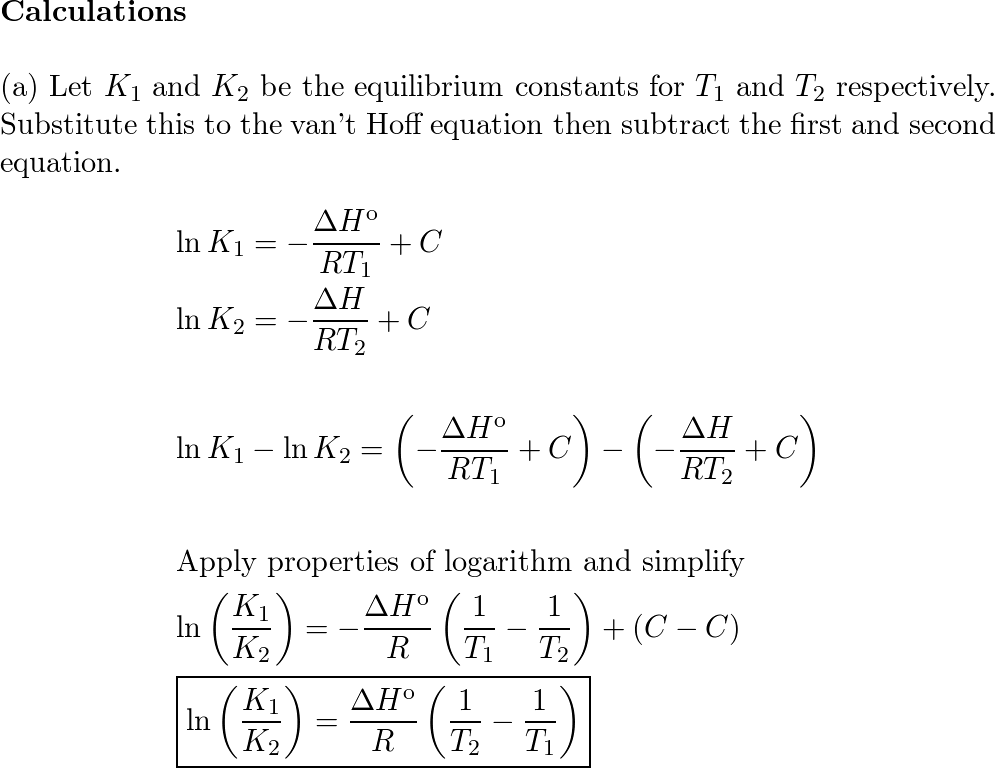PDF) Van't Hoff equation-equilibrium How much Van't Hoff equation -effect on temperature | Kajal Panda - Academia.edu

Lesson 19-van't hoff equation - Vant Hoff Equation The vant Hoff equation can be used to estimate Kc or Kp at a given temperature, provided that Kc or | Course Hero

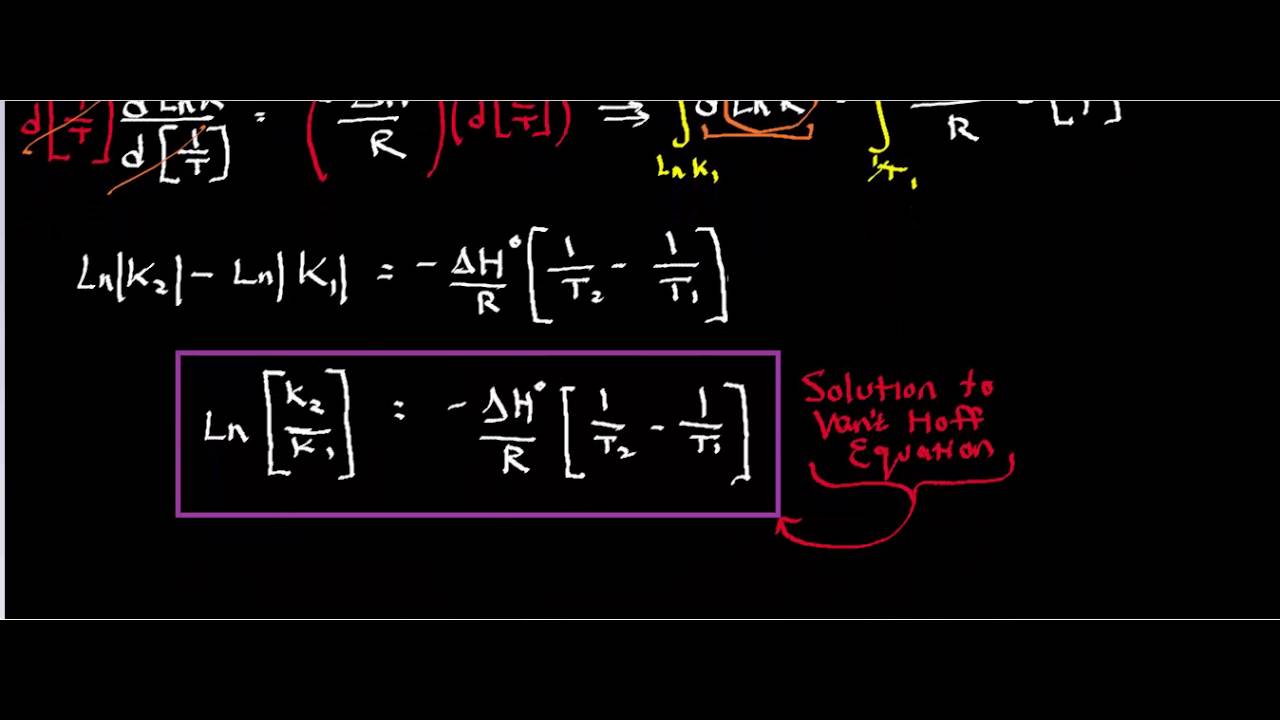
van't Hoff's equation gives the quantitative relation between change in value of K with change in temperature.

Van't Hoff reaction isochore : derivation and experimental determination of Kp at different temp. - YouTube