Transparency advocates win victory for public access to clinical trial data | Center for Science in the Public Interest

Vague FDA policies on adverse event data are keeping patients from accessing investigational drugs | Fierce Healthcare

Investigational New Drugs: FDA Has Taken Steps to Improve the Expanded Access Program but Should Further Clarify How Adverse Events Data Are Used | U.S. GAO

Lumma is FDA registered. I saw they updated their website i week ago stating they were, and i went to the FDA site and they are. Link: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfRL/rl.cfm : r/menstrualcups
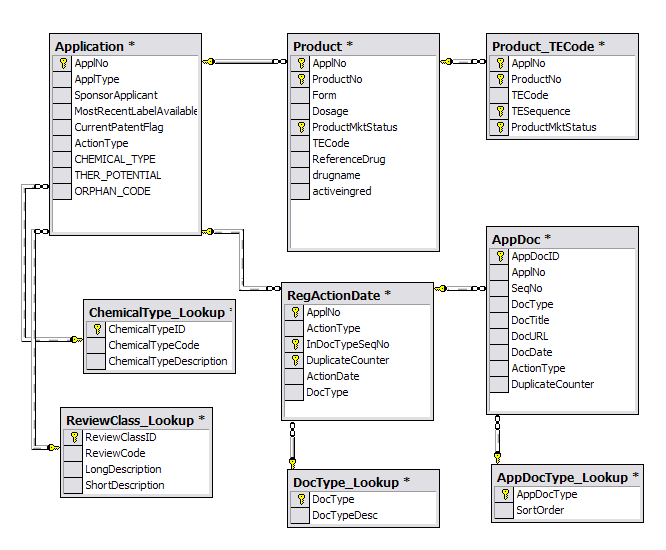
![PDF] IDAAPM: Integrated database of ADMET and adverse effects of predictive modeling based on FDA approved drug data PDF] IDAAPM: Integrated database of ADMET and adverse effects of predictive modeling based on FDA approved drug data](https://i1.rgstatic.net/publication/303977127_IDAAPM_Integrated_database_of_ADMET_and_adverse_effects_of_predictive_modeling_based_on_FDA_approved_drug_data/links/5900b6774585156502a00f65/largepreview.png)
PDF] IDAAPM: Integrated database of ADMET and adverse effects of predictive modeling based on FDA approved drug data

How Often Are Drugs Made Available Under the Food and Drug Administration's Expanded Access Process Approved? - McKee - 2017 - The Journal of Clinical Pharmacology - Wiley Online Library